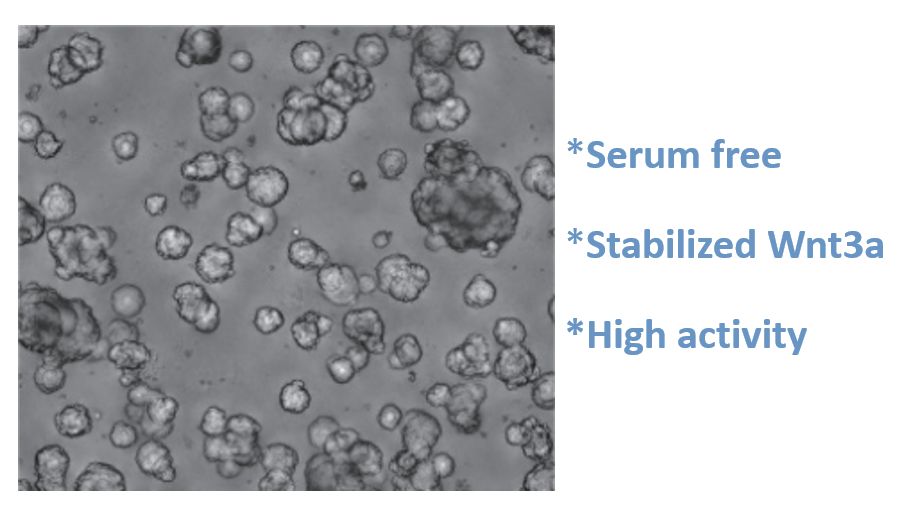
Afamin/Wnt3a Condition Medium
Condition medium to help organoid formation
Why the Wnt signaling pathway is important
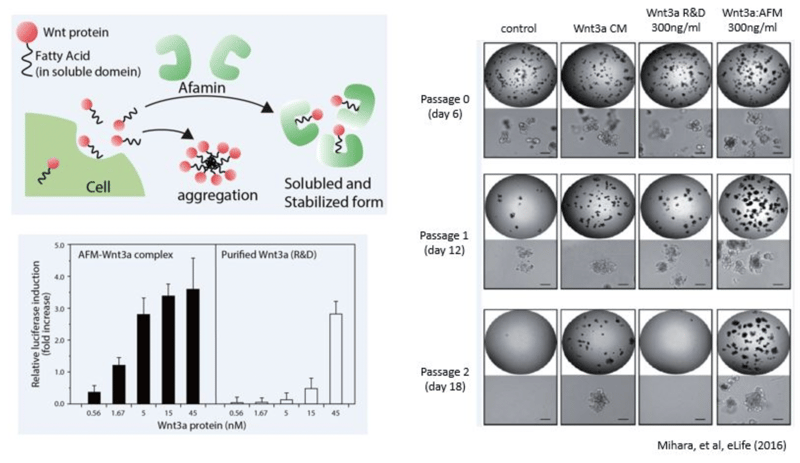
Wnt signaling is known to be involved in early development, maintenance and regeneration of stem cells, and in cancer formation. Wnt signaling has also been found to play an important role in the growth and maintenance of these processes. In particular, Wnt3a has been revealed to be an essential niche component for maintaining the proliferation of Lgr5-positive stem cells in intestinal epithelial cells and is used for the production of various digestive organoids such as the small intestine, large intestine, stomach, pancreas and liver. Although Wnt3a has been conventionally used for the culture of gut organoids, it is a fat-soluble protein, so it forms aggregates in serum-free medium and can not exert its activity sufficiently. In 2016, Mihara et al. found that high Wnt3a activity can be maintained by forming a complex with Wnt3a by Afamin, which is one of the components of serum. In addition, by using Afamin and Wnt3a complex for organoid culture, long-term culture of organoid becomes possible. This new medium will result in optimal success for your organoid experiments.
Mechanism of Wnt3a Stabilization by Afamin Proteins
Afamin/Wnt3a CM increased Lgr5 Positive Stem Cells Expressed td Tomato
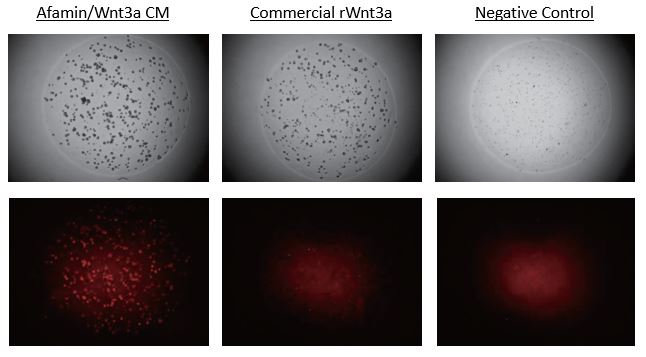
This data obtained from collaboration with Dr. Sato, Keio Univ
Images on the top panels show a bright field of human color organoids. Images on the bottom panels show fluorescent LGR5 positive stem cells that express td tomato regulated by Lgr5 promoter. Afamin/Wnt3a CM maintained LGR5 positive stem cell growth is seen at greater levels compared with cell growth in commercial recombinant Wnt3a (300ng/mL).
Recombinant Afamin/Wnt3a increased LGR5 Positive Stem Cells
An organoid strain expressing tdTomato regulated by Lgr5 promoter was cultured in the presence of Recombinant Afamin/Wnt3a and Recombinant Wnt3a of competitor A. It was confirmed that Recombinant Afamin/Wnt3a maintains LGR5-positive cells even at low concentrations.

What are the Differences Between Organoids and Spheroids?
In general, there are two major differences:
- Nature of the driving force for development: Whereas internal developmental processes drive organoid formation, spheroids develop primarily via cell-to-cell adhesion.
- Length of time 3D cultures can be maintained: Long term, in vitro expansion of cells in culture needs an immature stem cell population to replenish dying cells. Organoids are derived from, and maintain, a population of stem cells during in vitro culture, which guarantees their long term viability. This is achieved by optimizing culture growth conditions, such as providing a basement membrane matrix (i.e. Matrigel®), and adding a selection of agonists (e.g. Wnt and tyrosine kinase receptor) and inhibitors (e.g. bone morphogenetic protein/transforming growth factor-β).
Importantly, when organoids are passaged they retain the genetic features of the original organ over several generations. In contrast, long-term culture of tissue-derived spheroids is challenging, possibly due to inherent technical difficulties in extracting and maintaining viable cells.
Organoids and spheroids can be generated from a variety of healthy as well as diseased cell types and tissues, such as patient tumors. Tumor derived organoids and spheroids have been generated and extensively investigated for their use in drug discovery. However, there are some key differences in establishing the two from patient-derived tumors.
FGF-Max is our new product, shown to provide success with organoid cultures being acting as a universal FGF receptor agonist. Find out how this reagent works!
Product Highlights
References
- K. Ikezawa, et al., Establishment of organoids using residual samples from saline flushes during endoscopic ultrasound-guided fine-needle aspiration in patients with pancreatic cancer., Endosc Int Open 10 (2022) [PMID: 35036290]
- K. Toshimitsu, et al., Organoid screening reveals epigenetic vulnerabilities in human colorectal cancer., Nat Chem Biol 18 (2022) [PMID: 35273398]
- Z. Wang, et al., Lactate promotes the growth of patient-derived organoids from hepatopancreatobiliary cancers via ENO1/HIF1α pathway and does not affect their drug sensitivities., Cell Death Discov 8 (2022) [PMID: 35443744]
- C. Cao, et al., Phenotypical screening on metastatic PRCC-TFE3 fusion translocation renal cell carcinoma organoids reveals potential therapeutic agents., Clin Transl Oncol (2022) [PMID: 35118587]
- K. Togasaki, et al., Wnt Signaling Shapes the Histologic Variation in Diffuse Gastric Cancer., Gastroenterology 160 (2021) [PMID: 33217450]
- S. Sugimoto, et al., An organoid-based organ-repurposing approach to treat short bowel syndrome., Nature 592 (2021) [PMID: 33627870]
- T. De Oliveira, et al., Effects of the Novel PFKFB3 Inhibitor KAN0438757 on Colorectal Cancer Cells and Its Systemic Toxicity Evaluation In Vivo., Cancers (Basel) 13 (2021) [PMID: 33671096]
- T. Nishina, et al., Interleukin-11-expressing fibroblasts have a unique gene signature correlated with poor prognosis of colorectal cancer., Nat Commun 12 (2021) [PMID: 33863879]
- T. Ebisudani, et al., Direct derivation of human alveolospheres for SARS-CoV-2 infection modeling and drug screening., Cell Rep 35 (2021) [PMID: 34038715]
- D. Kim, et al., 3D Organoid Culture From Adult Salivary Gland Tissues as an ex vivo Modeling of Salivary Gland Morphogenesis., Front Cell Dev Biol 9 (2021) [PMID: 34458260]
- EA. Farshadi, et al., Organoids Derived from Neoadjuvant FOLFIRINOX Patients Recapitulate Therapy Resistance in Pancreatic Ductal Adenocarcinoma., Clin Cancer Res 27 (2021) [PMID: 34580113]
- YW. Cho, et al., Patient-derived organoids as a preclinical platform for precision medicine in colorectal cancer., Mol Oncol (2021) [PMID: 34850547]
- S. Sugimoto, et al., Organoid Derivation and Orthotopic Xenotransplantation for Studying Human Intestinal Stem Cell Dynamics., Methods Mol Biol 2171 (2020) [PMID: 32705652]
- K. Nanki, et al., Somatic inflammatory gene mutations in human ulcerative colitis epithelium., Nature 577 (2020) [PMID: 31853059]
- N. Sasaki, et al., Development of a Scalable Coculture System for Gut Anaerobes and Human Colon Epithelium., Gastroenterology 159 (2020) [PMID: 32199883]
- S. Mae, et al., Expansion of Human iPSC-Derived Ureteric Bud Organoids with Repeated Branching Potential., Cell Reports 32 (2020) [PMID: 32726627]
- Y. Nanki, et al., Patient-derived ovarian cancer organoids capture the genomic profiles of primary tumours applicable for drug sensitivity and resistance testing., Scientific Reports 28 (2020) [PMID: 32724113]
- K. Miyabayashi, et al., Intraductal transplantation models of human pancreatic ductal adenocarcinoma reveal progressive transition of molecular subtypes., Cancer Discov 10 (2020) [PMID: 32703770]
- K. Kawasaki, et al., Chromosome Engineering of Human Colon-Derived Organoids to Develop a Model of Traditional Serrated Adenoma., Gastroenterology 158 (2020) [PMID: 31622618]
- K. Kawasaki, et al., An Organoid Biobank of Neuroendocrine Neoplasms Enables Genotype-Phenotype Mapping., Cell 183 (2020) [PMID: 33159857]
- H. Oshima, et al., Stat3 is indispensable for damage-induced crypt regeneration but not for Wnt-driven intestinal tumorigenesis., FASEB J 33 (2019) [PMID: 30156908]
- K. Nanki, et al., Divergent routes toward Wnt and R-spondin niche independency during human gastric carcinogenesis., Cell 174 (2018) [PMID: 30096312]
- S. Sugimoto, et al., Reconstruction of the human colon epithelium in vivo., Cell Stem Cell 22 (2018) [PMID: 29290616]
- T. Seino, et al., Human pancreatic tumor organoids reveal loss of stem cell niche factor dependence during disease progression., Cell Stem Cell 22 (2018) [PMID: 29337182]
- H. Tiriac, et al., Successful creation of pancreatic cancer organoids by means of EUS-guided fine-needle biopsy sampling for personalized cancer treatment., Gastrointest Endosc 87 (2018) [PMID: 29325707]
- H. Tiriac, et al., Organoid Profiling Identifies Common Responders to Chemotherapy in Pancreatic Cancer., Cancer Discov 8 (2018) [PMID: 29853643]
- M. Fujii, et al., Human Intestinal Organoids Maintain Self-Renewal Capacity and Cellular Diversity in Niche-Inspired Culture Condition., Cell Stem Cell 23 (2018) [PMID: 30526881]
- JS. Roe, et al., Enhancer Reprogramming Promotes Pancreatic Cancer Metastasis., Cell 170 (2017) [PMID: 28757253]
- E. Mihara, et al., Active and water-soluble form of lipidated Wnt protein is maintained by a serum glycoprotein afamin/α-albumin., eLife 5 (2016) [PMID: 26902720]


